CONJUGATE ACID-BASE PAIRS Name
advertisement
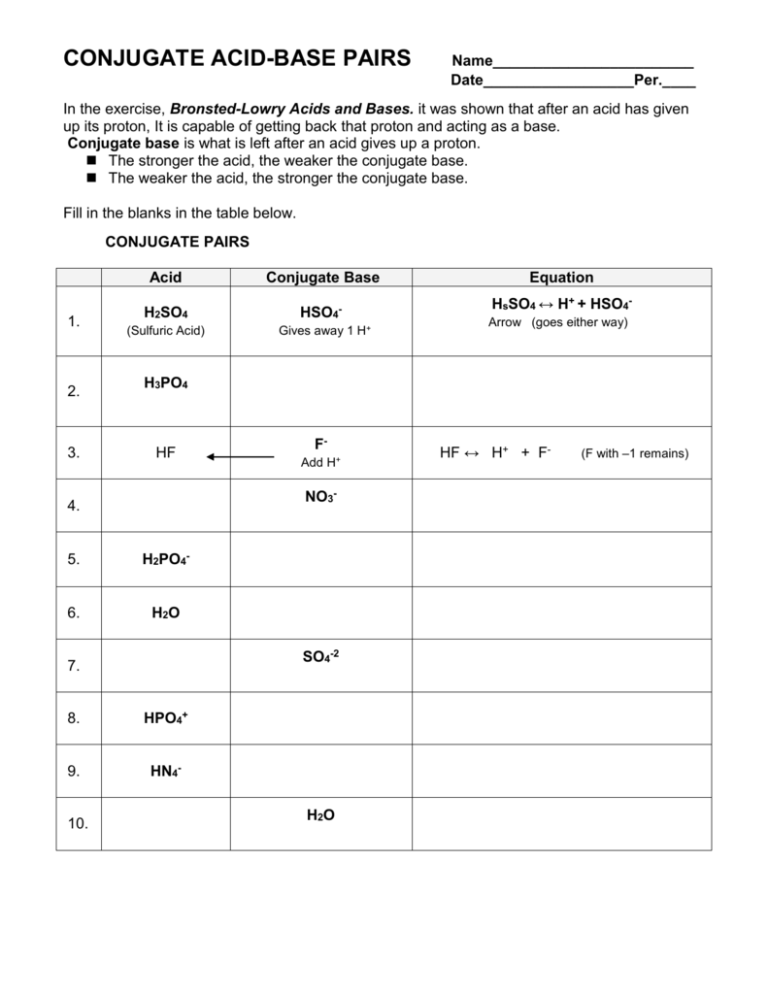
CONJUGATE ACID-BASE PAIRS Name________________________ Date__________________Per.____ In the exercise, Bronsted-Lowry Acids and Bases. it was shown that after an acid has given up its proton, It is capable of getting back that proton and acting as a base. Conjugate base is what is left after an acid gives up a proton. The stronger the acid, the weaker the conjugate base. The weaker the acid, the stronger the conjugate base. Fill in the blanks in the table below. CONJUGATE PAIRS 1. 2. 3. Acid Conjugate Base H2SO4 HSO4- (Sulfuric Acid) HsSO4 ↔ H+ + HSO4Arrow (goes either way) H3PO4 HF FAdd H+ NO3- 4. 5. H2PO4- 6. H2O SO4-2 7. 8. HPO4+ 9. HN4- 10. Gives away 1 H+ Equation H2O HF ↔ H+ + F- (F with –1 remains) Name _____________________ Date _____________Per ______ BRONSTED - LOWRY ACIDS AND BASES According to Bronsted-Lowry theory, an acid is a proton (H+) donor, and a base is a proton acceptor. H+ Example: HCI + OH- → Cl- + H2O Acid give away H+ The HCI acts as an acid, the OH as a base H2O (acid) This reaction is reversible in that the H2O Cl- (base) can give back the proton to the Cl Label the Bronsted-Lowry acids and bases in the following reactions and show the direction of proton transfer. H+ H- H2O gave H(Acid) + Cl picks up H (Base) [now conjugate Acid] Example: H2O + Cl- ↔ OH- + HCl Acid Base C. Base C. Acid 1. H20 + H20 Acid ↔ Base H3O+ C. Acid + OHC. Base 2. H2S04 + OH- ↔ HSO4 - + H2O+ 3. HSO4 - + H2O ↔ SO4-2 + H3O+ 4. OH- + H3O+ ↔ H2O + H20 5. NH3 + H2O ↔ NH4+ + OH-











