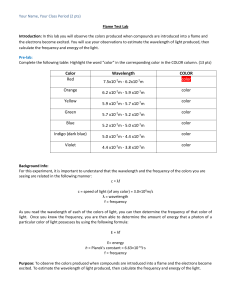
Flame Test Lab
8th Grade Science
Mrs. Dainty
Review
Can we define the following terms?
Atom
Element
Metal
Compound
Bunsen Burner
Physical Characteristic
Chemical Characteristic
Energy Levels
Light
Background
When certain metal compounds, or metal salts, are
heated, their electrons enter an excited state.
In this excited state, electrons move from lower energy
levels to higher energy levels.
This excited state is unstable, so the electrons quickly
return to their lower energy levels. When this happens,
the lose energy in the form of LIGHT.
When this light is in the visible range, we can see it.
(Remember Roy G. Biv!)
Background
Flame tests can be conducted to determine the identity of
unknown elements.
Certain elements give off known colors of light. These can
be compared with the color of light given off by an
unknown substance.
Your Job:
Your mission is to discover what metal elements are
present in a dehydrated banana by conducting a flame
test.
First, you will discover the colors given off by:
Calcium Chloride
Copper Chloride
Lithium Chloride
Potassium Chloride
Sodium Chloride
Strontium Chloride
Then, you will test a banana to see if any color change
occurs in the flame. Compare these results to the known
elements to draw conclusions about what’s in the banana!
Limitations
The flame test may not detect atoms that are present in
low concentrations.
The flame test cannot distinguish between ALL elements.
(Some elements create the same color. Some elements
don’t create a visible change in the flame color at all.)
Impurities and contaminates can affect your results.
Science Notebook
In your notebook, write a title and date for this lab.
Next, write the question we’re investigating in your own
words. Your questions should make it very clear what we
are trying to learn, and should identify any variables being
tested.
Finally, make a prediction. (I think that…because…)
Demonstration
Flame test demonstration:
1st time – watch and listen
2nd time – write a materials list and procedures
Safety Considerations
Utmost care must be taken during this lab. Absolutely no goofing
around will be tolerated.
Minimize movement around the room, as well as talking with your
hands! (Materials Manger only one to walk around to get supplies.)
Goggles, gloves, and lab coats are to be worn.
Long hair must be tied back.
Know locations of emergency equipments: eye wash, shower, fire
blanket, fire extinguishers, etc.
Voices should be kept low.
In case of emergency, the FIRST thing you do is to find your teacher!
Follow instructions for disposal of chemicals.
Science Notebook
Once you have written down the materials and
procedures, develop a table in your results section.
You must have your table ready before you can begin the
lab.
Chemicals you’ll test:
Calcium Chloride
Copper Chloride
Lithium Chloride
Potassium Chloride
Sodium Chloride
Strontium Chloride
Unknown (Banana)
Lab Day
Lab Groups:
Get goggles, gloves, and lab
coats.
Place books at tables near
sinks. Bring ONLY lab
notebooks to work station at
counter.
Go to your designated lab
station. Clear away unneeded
materials. Designate group
roles. (If only 3, share
spokesperson responsibilities.)
Wait for permission to
begin.
Non-Lab Groups:
Get goggles.
Choose a place to sit near the
windows. DO NOT SIT BY
ANYONE WHO WILL TEMPT
YOU TO GOOF OFF. My
attention needs to mainly be
with lab groups and I will have
little patience for goofing
around today!
Work: Ch. 3, Lesson 4
If finished, can take turns
“building an element.”
(Further instructions to come.)
 0
0