Chapter 5
Section 4
Objectives
•
Describe how carbon atoms bond covalently to form organic compounds.
•
Identify the names and structures of groups of simple organic compounds and
polymers.
•
Identify what makes up the polymers that are essential to life.
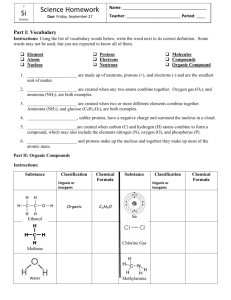
Organic Compounds
•
In chemistry, the word organic is used to describe certain compounds.
•
An organic compound is a covalently bonded compound that contains carbon,
excluding carbonates and oxides.
•
Many ingredients of familiar substances
contain carbon.
•
Carbon atoms form four covalent bonds in
organic compounds.
•
When a compound is made of only carbon and hydrogen atoms, it is called a
hydrocarbon.
•
Alkanes are hydrocarbons that have only single covalent bonds.
•
•
Examples:
The carbon atoms in any alkane with more than
three carbon atoms can have more than one
possible arrangement.
•
Carbon atom chains may be branched or unbranched, and they can even form
rings.
•
Except for cyclic alkanes, the chemical formulas for alkanes follow a special
pattern. The number of hydrogen atoms is always two more than twice the
number of carbon atoms.
•
Alkenes are hydrocarbons that contain double carbon-carbon bonds.
•
•
Alcohols have hydroxyl, or –OH, groups.
•
•
Example: polyethene (often known as polyethylene) is a long chain made
from many molecules of ethene.
Some polymers are natural; others are man-made.
•
•
Alcohols, which have the suffix -ol in their names, are found in many
household products.
A polymer is large molecule that is formed by more than five monomers, or small
units.
•
•
Example: methanol, CH3OH
Alcohol molecules behave similarly to water molecules.
•
•
Example: ethene,
Examples: rubber, starch, protein, and DNA are all natural polymers.
Plastics and synthetic fibers are man-made polymers.
The elasticity of a polymer is determined by
its structure.
•
Examples:
•
A milk jug made of polyethene is not elastic: it
can be crushed, but does not return to its
original shape.
•
A rubber band is an elastic polymer: when it
is stretched and released, it returns to its
original shape.
Biochemical Compounds
•
A carbohydate is any organic compound that is made of carbon, hydrogen, and
oxygen and that provides nutrients to the cells of living things.
•
A protein is an organic compound that is a polymer of amino acids, and a
principal component of all cells.
•
An amino acid is any one of 20 different organic molecules that contain a
carboxyl and an
amino group.
•
Your DNA determines your entire genetic makeup.
•
DNA is a polymer with a complex structure. It is in the form of paired strands, in
the shape of a twisted ladder known as a double helix.
•
Each time a new cell is made in your body, a new copy of your DNA is made for
the new cell.
•
The two strands in the helix are separated each time your DNA is copied.
 0
0