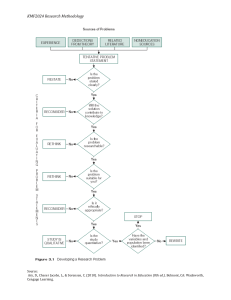
62
chapter 3 Organic Compounds: Alkanes and Their Stereochemistry
Table 3.1 Structures of Some Common Functional Groups
Name
Structurea
Alkene
(double bond)
C
Alkyne
(triple bond)
Name ending
Example
-ene
H2CPCH2
Ethene
-yne
HCqCH
Ethyne
C
OCqCO
Arene
(aromatic ring)
None
Benzene
Halide
C
X
None
CH3Cl
Chloromethane
-ol
CH3OH
Methanol
ether
CH3OCH3
Dimethyl ether
(X F, Cl, Br, I)
Alcohol
C
Ether
C
OH
O
Monophosphate
C
O
C
O
P
O–
C
O
P
O–
Amine
C
Imine
(Schiff base)
Nitrile
Thiol
C
OCqN
C
O
diphosphate
CH3OP2O63
Methyl diphosphate
O
P
O–
O–
-amine
CH3NH2
Methylamine
None
NH
N
N
C
CH3OPO32
Methyl phosphate
O–
O
Diphosphate
phosphate
SH
CH3CCH3
C
Acetone imine
-nitrile
CH3CqN
Ethanenitrile
-thiol
CH3SH
Methanethiol
aThe bonds whose connections aren’t specified are assumed to be attached to carbon or hydrogen atoms in the rest of the molecule.
(Continued)
Unless otherwise noted, all content on this page is © Cengage Learning.
Copyright 2015 Cengage Learning. All Rights Reserved. May not be copied, scanned, or duplicated, in whole or in part. Due to electronic rights, some third party content may be suppressed from the eBook and/or eChapter(s).
Editorial review has deemed that any suppressed content does not materially affect the overall learning experience. Cengage Learning reserves the right to remove additional content at any time if subsequent rights restrictions require it.
3-1 Functional Groups
Table 3.1 Structures of Some Common Functional Groups continued
Name
Sulfide
Structurea
C
Disulfide
C
S
C
S
C
O–
Sulfoxide
C
Aldehyde
S
Name ending
S+
Ketone
Carboxylic acid
Ester
Thioester
Amide
Acid chloride
C
C
C
C
C
C
Ethanoic acid
-oate
C
C
Methyl ethanoate
O
CH3CSCH3
C
Methyl ethanethioate
-amide
O
CH3CNH2
N
Ethanamide
-oyl chloride
O
CH3CCl
Cl
Ethanoyl chloride
O
O
O
CH3COCH3
C
-thioate
S
O
CH3COH
OH
O
O
Propanone
-oic acid
O
Carboxylic acid
anhydride
O
CH3CCH3
C
O
C
O–
+
CH3SCH3
-one
O
C
sulfoxide
CH3CH
Ethanal
O
C
CH3SSCH3
Dimethyl disulfide
H
O
C
disulfide
-al
O
C
CH3SCH3
Dimethyl sulfide
Dimethyl sulfoxide
O
C
sulfide
C
O
C
Example
C
-oic
anhydride
C
O O
CH3COCCH3
Ethanoic anhydride
aThe bonds whose connections aren’t specified are assumed to be attached to carbon or hydrogen atoms in the rest of the molecule.
Unless otherwise noted, all content on this page is © Cengage Learning.
Copyright 2015 Cengage Learning. All Rights Reserved. May not be copied, scanned, or duplicated, in whole or in part. Due to electronic rights, some third party content may be suppressed from the eBook and/or eChapter(s).
Editorial review has deemed that any suppressed content does not materially affect the overall learning experience. Cengage Learning reserves the right to remove additional content at any time if subsequent rights restrictions require it.
63
 0
0