The manufacture, sale, and distribution of chemical products is one of the cornerstones of a
developed country. Chemists play an important role in the manufacture, inspection, and safe
handling of chemical products, as well as in product development and general management.
The manufacture of basic chemicals such as oxygen, chlorine, ammonia, and sulfuric
acid provides the raw materials for industries producing textiles, agricultural products, metals,
paints, and pulp and paper. Specialty chemicals are produced in smaller amounts for industries
involved with such products as pharmaceuticals, foodstuffs, packaging, detergents, flavours, and
fragrances. To a large extent, the chemical industry takes the products and reactions common to
“bench-top” chemical processes and scales them up to industrial quantities.
The monitoring and control of bulk chemical processes, especially with regard to heat transfer,
pose problems usually tackled by chemists and chemical engineers. The disposal of by-products
also is a major problem for bulk chemical producers. These and other challenges of industrial
chemistry set it apart from the more purely intellectual disciplines of chemistry discussed
above. Yet, within the chemical industry, there is a considerable amount of fundamental
research undertaken within traditional specialties. Most large chemical companies have
research-and-development capability. Pharmaceutical firms, for example, operate large research
laboratories in which chemists test molecules for pharmacological activity. The new products
and processes that are discovered in such laboratories are often patented and become a source
of profit for the company funding the research. A great deal of the research conducted in the
chemical industry can be termed applied research because its goals are closely tied to the
products and processes of the company concerned. New technologies often require much
chemical expertise. The fabrication of, say, electronic microcircuits involves close to 100
separate chemical steps from start to finish. Thus, the chemical industry evolves with the
technological advances of the modern world and at the same time often contributes to the rate
of progress.
The methodology of chemistry
molecular structureA ball-and-stick model of molecular structure, showing atoms bonded
together.
Chemistry is to a large extent a cumulative science. Over time the number and extent of
observations and phenomena studied increase. Not all hypotheses and discoveries endure
unchallenged, however. Some of them are discarded as new observations or more satisfying
explanations appear. Nonetheless, chemistry has a broad spectrum of explanatory models for
chemical phenomena that have endured and been extended over time. These now have the
status of theories, interconnected sets of explanatory devices that correlate well with observed
phenomena. As new discoveries are made, they are incorporated into existing theory whenever
possible. However, as the discovery of high-temperature superconductors in 1986 illustrates,
accepted theory is never sufficient to predict the course of future discovery. Serendipity, or
chance discovery, will continue to play as much a role in the future as will theoretical
sophistication.
Studies of molecular structure
The chemical properties of a substance are a function of its structure, and the techniques of Xray crystallography now enable chemists to determine the precise atomic arrangement of
complex molecules. A molecule is an ordered assembly of atoms. Each atom in a molecule is
connected to one or more neighbouring atoms by a chemical bond. The length of bonds and the
angles between adjacent bonds are all important in describing molecular structure, and
a comprehensive theory of chemical bonding is one of the major achievements of modern
chemistry. Fundamental to bonding theory is the atomic–molecular concept.
Atoms and elements
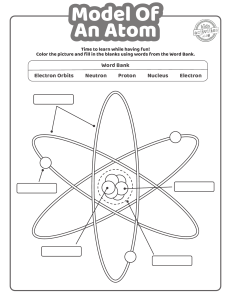
As far as general chemistry is concerned, atoms are composed of the three fundamental
particles: the proton, the neutron, and the electron. Although the proton and the neutron are
themselves composed of smaller units, their substructure has little impact on chemical
transformation. As was explained in an earlier section, the proton carries a charge of +1, and the
number of protons in an atomic nucleus distinguishes one type of chemical atom from another.
The simplest atom of all, hydrogen, has a nucleus composed of a single proton. The neutron has
very nearly the same mass as the proton, but it has no charge. Neutrons are contained with
protons in the nucleus of all atoms other than hydrogen. The atom with one proton and one
neutron in its nucleus is called deuterium. Because it has only one proton, deuterium exhibits
the same chemical properties as hydrogen but has a different mass. Hydrogen and deuterium
are examples of related atoms called isotopes. The third atomic particle, the electron, has a
charge of -1, but its mass is 1,836 times smaller than that of a proton. The electron occupies a
region of space outside the nucleus termed an orbital. Some orbitals are spherical with the
nucleus at the centre. Because electrons have so little mass and move about at speeds close to
half that of light, they exhibit the same wave–particle duality as photons of light. This means
that some of the properties of an electron are best described by considering the electron to be
a particle, while other properties are consistent with the behaviour of a standing wave.
The energy of a standing wave, such as a vibrating string, is distributed over the region of space
defined by the two fixed ends and the up-and-down extremes of vibration. Such a wave does
not exist in a fixed region of space as does a particle. Early models of atomic
structure envisioned the electron as a particle orbiting the nucleus, but electron orbitals are
now interpreted as the regions of space occupied by standing waves called wave functions.
These wave functions represent the regions of space around the nucleus in which the
probability of finding an electron is high. They play an important role in bonding theory, as will
be discussed later.
Each proton in an atomic nucleus requires an electron for electrical neutrality. Thus, as the
number of protons in a nucleus increases, so too does the number of electrons. The electrons,
alone or in pairs, occupy orbitals increasingly distant from the nucleus. Electrons farther from
the nucleus are attracted less strongly by the protons in the nucleus, and they can be removed
more easily from the atom. The energy required to move an electron from one orbital to
another, or from one orbital to free space, gives a measure of the energy level of the orbitals.
These energies have been found to have distinct, fixed values; they are said to be quantized. The
energy differences between orbitals give rise to the characteristic patterns of light absorption or
emission that are unique to each chemical atom.
A new chemical atom—that is, an element—results each time another proton is added to an
atomic nucleus. Consecutive addition of protons generates the whole range of elements known
to exist in the universe. Compounds are formed when two or more different elements combine
through atomic bonding. Such bond formation is a consequence of electron pairing
and constitutes the foundation of all structural chemistry.
 0
0