Equations of State
Compiled by:
Gan Chin Heng / Shermon Ong
07S06G / 07S06H
How are states represented?
Diagrammatically (Phase diagrams)
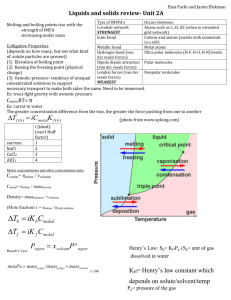
Pressure
Solid
Liquid
Critical point
Triple point
Gas
Temp
How are states represented?
Mathematically
Using
equations of state
Relate state variables to describe property of
matter
Examples of state variables
Pressure
Volume
Temperature
Equations of state
Mainly used to describe fluids
Liquids
Gases
Particular emphasis today on gases
ABCs of gas equations
Law
B
Boyle’s Law
C
Charles’ Law
A
Avogadro’s
Avogadro’s Law
At constant temperature and pressure
Volume
of gas proportionate to amount of gas
i.e. V n
Independent of gas’ identity
Approximate molar volumes of gas
dm3 at 298K
22.4 dm3 at 273K
24.0
Boyle’s Law
At constant temperature
and amounts
Gas’
volume inversely
proportionate to pressure,
i.e. V 1/p
The product of V & p, which
is constant, increases with
temperature
Charles’ Law
At constant pressure
and amounts
Volume
proportionate to
temperature, i.e. V T
T
is in Kelvins
Note the extrapolated
lines (to be explained
later)
Combining all 3 laws…
V (1/p)(T)(n)
V nT/p
Rearranging, pV = (constant)nT
Thus we get the ideal gas equation:
pV = nRT
Assumptions
Ideal gas particles occupy
negligible volume
Ideal gas particles have
negligible intermolecular
interactions
But sadly assumptions
fail…Nothing is ideal in this world…
It’s downright
squeezy here
Real gas particles DO occupy
finite volume
Real gas particles have
considerable intermolecular
interactions
Failures of ideal gas equation
Failure of Charles’ Law
At
very low
temperatures
Volume do not
decrease to zero
Gas liquefies instead
Remember the
extrapolated lines?
Failures of ideal gas equation
From pV = nRT, let Vm be molar volume
pVm
= RT
pVm / RT = 1
pVm / RT is also known as Z, the
compressibility factor
Z should be 1 at all conditions for an ideal
gas
Failures of ideal gas equation
Looking at Z plot
of real gases…
Obvious deviation
from the line Z=1
Failure of ideal gas
equation to
account for these
deviations
So how?
A Dutch physicist named Johannes Diderik
van der Waals devised a way...
Johannes Diderik van der Waals
November 23, 1837
– March 8, 1923
Dutch
1910 Nobel Prize in
Physics
So in 1873…
I can approximate
the behaviour of
fluids with an
equation
Scientific
community
ORLY?
YARLY!
Van der Waals Equation
Modified from ideal gas equation
Accounts for:
Non-zero
volumes of gas particles (repulsive
effect)
Attractive forces between gas particles
(attractive effect)
Van der Waals Equation
Attractive effect
Pressure
= Force per unit area of container
exerted by gas molecules
Dependent on:
Frequency of collision
Force of each collision
Both
factors affected by attractive forces
Each factor dependent on concentration (n/V)
Van der Waals Equation
Hence
pressure changed proportional to
(n/V)2
Letting a be the constant relating p and
(n/V)2…
Pressure term, p, in ideal gas equation
becomes [p+a(n/V)2]
Van der Waals Equation
Repulsive effect
Gas
molecules behave like small,
impenetrable spheres
Actual volume available for gas smaller than
volume of container, V
Reduction in volume proportional to amount of
gas, n
Van der Waals Equation
Let
another constant, b, relate amount of gas,
n, to reduction in volume
Volume term in ideal gas equation, V,
becomes (V-nb)
Van der Waals Equation
Combining both derivations…
We get the Van der Waals Equation
2
n
p + a [V-nb] = nRT
V
OR
a
p + 2 [Vm -b] = RT
Vm
Van der Waals Equation -> So
what’s the big deal?
Real world significances
Constants
a and b depend on the gas identity
Relative values of a and b can give a rough
comparison of properties of both gases
Van der Waals Equation -> So
what’s the big deal?
Value of constant a
Gives
a rough indication of magnitude of
intermolecular attraction
Usually, the stronger the attractive forces, the higher
is the value of a
Some values (L2 bar mol-2):
Water: 5.536
HCl: 3.716
Neon: 0.2135
Van der Waals Equation -> So
what’s the big deal?
Value of constant b
Gives
a rough indication of size of gas molecules
Usually, the bigger the gas molecules, the higher is
the value of b
Some values (L mol-1):
Benzene: 0.1154
Ethane: 0.0638
Helium: 0.0237
Critical temperature and
associated constants
Critical temperature?
Given a p-V plot of a
real gas…
At higher temperatures
T3 and T4, isotherm
resembles that of an
ideal gas
Critical temperature?
At T1 and V1, when gas volume
decreased, pressure increases
From V2 to V3, no change in
pressure even though volume
decreases
Condensation taking place and
pressure = vapor pressure at T1
Pressure rises steeply after V3
because liquid compression is
difficult
Critical temperature?
At higher temperature T2,
plateau region becomes shorter
At a temperature Tc, this
‘plateau’ becomes a point
Tc is the critical temperature
Volume at that point, Vc =
critical volume
Pressure at that point, Pc =
critical pressure
Critical temperature
At T > Tc, gas can’t be compressed into
liquid
At Tc, isotherm in a p-V graph will have a
point of inflection
1st
and 2nd derivative of isotherm = 0
We shall look at a gas obeying the Van der
Waals equation
VDW equation and critical
constants
Using VDW equation,
we can derive the
following
a
p + 2 [Vm -b] = RT
Vm
RT
a
p=
- 2
Vm -b Vm
VDW equation and critical
constants
At Tc, Vc and Pc, it’s a
point of inflexion on pVm graph
dp
0
dVm T
d p
0
2
dVm T
2
VDW equation and critical
constants
dp
RT
2a
3
2
(Vm b) Vm
dVm T
d2 p
2 RT
6a
4
2
3
dVm T (Vm b) Vm
Rearranging...
a
8a
Vm,c = 3b; p c =
; Tc =
2
27b
27Rb
p c Vm,c
3
Zc =
=
RTc
8
VDW equation and critical
constants
Qualitative trends
As
seen from formula, bigger molecules decrease
critical temperature
Stronger IMF increase critical temperature
Usually outweighs size factor as bigger molecules have
greater id-id interaction
Real values:
Water: 647K
Oxygen: 154.6K
Neon: 44.4K
Helium: 5.19K
Compressibility Factor
Compressibility Factor
Recall Z plot?
Z = pVm / RT; also
called the
compressibility factor
Z should be 1 at all
conditions for an ideal
gas
Compressibility Factor
For real gases, Z not
equals to 1
Z = Vm / Vm,id
Implications:
At
high p, Vm > Vm,id,
Z>1
Repulsive forces
dominant
Compressibility Factor
At
intermediate p, Z <
1
Attractive forces
dominant
More significant for
gases with significant
IMF
Boyle Temperature
Z also varies with temperature
At a particular temperature
Z
= 1 over a wide range of pressures
That means gas behaves ideally
Obeys Boyle’s Law (recall V 1/p)
This temperature is called Boyle Temperature
Boyle Temperature
Mathematical
implication
Initial gradient of Z-p plot = 0 at T
dZ/dp = 0
For
a gas obeying VDW equation
TB = a / Rb
Low Boyle Temperature favoured by weaker IMF
and bigger gas molecules
Virial Equations
Virial Equations
Recall compressibility factor Z?
Z
= pVm/RT
Z = 1 for ideal gases
What about real gases?
Obviously
Z≠1
So how do virial equations address this
problem?
Virial Equations
Form
= 1 + B/Vm + C/Vm2 + D/Vm3 + …
pVm/RT = 1 + B’p + C’p2 + D’p3 + …
pVm/RT
B,B’,C,C’,D & D’ are virial coefficients
Temperature
dependent
Can be derived theoretically or experimentally
Virial Equations
Most flexible form of state equation
Terms
can be added when necessary
Accuracy can be increase by adding infinite
terms
For same gas at same temperature
Coefficients
B and B’ are proportionate but not
equal to each other
Summary
Summary
States can be represented using diagrams or equations
Ideal Gas Equation combines Avagadro's, Boyle's and
Charles' Laws
Assumptions of Ideal Gas Equation fail for real gases,
causing deviations
Van der Waals Gas Equation accounts for attractive and
repulsive effects ignored by Ideal Gas Equation
Summary
Constants a and b represent the properties of a real gas
A gas with higher a value usually has stronger IMF
A gas with higher b value is usually bigger
A gas cannot be condensed into liquid at temperatures
higher than its critical temperature
Summary
Critical temperature is represented as a point of inflexion
on a p-V graph
Compressibility factor measures the deviation of a real
gas' behaviour from that of an ideal gas
Boyle Temperature is the temperature where Z=1 over a
wide range of pressures
Boyle Temperature can be found from Z-p graph where
dZ/dp=0
Summary
Virial equations are highly flexible
equations of state where extra terms can
be added
Virial equations' coefficients are
temperature dependent and can be
derived experimentally or theoretically
 0
0